Beer Contamination Prevention and Detection Using Aseptic Inline Sampling
In the brewing industry, ensuring the final product’s quality and safety is vital. Effective process monitoring and beer contamination control are crucial to maintaining product integrity. Inline sampling, a method that allows for real-time analysis of the brewing process, has emerged as a valuable tool for breweries. Aseptic inline sampling techniques can help breweries proactively identify and address potential issues, resulting in improved product consistency and reduced risk of contamination.
Understanding Beer Quality Control and Process Monitoring
To fully appreciate the significance of aseptic inline sampling, it is essential to understand the concept of process monitoring and the potential risks associated with contamination in breweries.
Process Monitoring encompasses systematically observing and measuring various parameters throughout the brewing process. This includes monitoring factors such as temperature, pH levels, yeast activity, and fermentation progress. By closely monitoring these variables, breweries can assess the health of the brewing process and make timely adjustments, if necessary.
Contamination refers to the unintended introduction of microorganisms or impurities into the brewing process. Contamination can enter your process from biofilms, cracks, pinholes, and other unwanted sources that can adversely affect the taste, aroma, and overall quality of the beer. Contamination can occur at various stages of the brewing process, including during raw material handling, fermentation, dry hopping, product transfer, and packaging.
The Role of Aseptic Inline Sampling in Breweries
Aseptic inline sampling involves the sterile collection of samples directly from the brewing process, providing critical information on key parameters. This technique allows breweries to gain immediate and accurate insights into the quality and consistency of their product, minimizing the risk of introducing contaminants into the process. The benefits of aseptic inline sampling extend beyond process monitoring; it also serves as a valuable tool for seltzer and beer contamination control.
Ensuring Water Purity with Aseptic Inline Sampling
Water is one of the most critical ingredients in brewing and beverage production. Aseptic inline sampling ensures that the water used throughout the process is free from contaminants, safeguarding the quality and safety of the final product. This proactive approach helps detect and address potential issues early on, reducing the risk of spoilage and ensuring product consistency.
Continuous monitoring with aseptic inline sampling provides real-time data on water quality. This capability is especially important for breweries and beverage manufacturers where even minor contamination can affect flavor profiles and product quality. By incorporating aseptic inline sampling into the production process, companies can maintain high standards of quality assurance and the purity of their seltzer and beer products.
Additional key advantages of implementing aseptic sampling in breweries are:
- Timely Detection of Quality Issues: Aseptic inline sampling enables breweries to identify and isolate issues within their process that may impact product quality. By continuously monitoring critical control points, breweries can quickly detect any abnormalities and promptly take corrective actions. This proactive approach minimizes the risk of producing off-flavored or spoiled beer.
- Improved Product Consistency: Aseptic inline sampling provides breweries with a greater level of control over the brewing process. By closely monitoring key parameters, breweries can ensure consistency from batch to batch, resulting in a more uniform and predictable product. This consistency is essential for establishing a strong brand reputation and meeting consumer expectations.
- Early Contamination Detection: Contamination can have severe consequences for breweries, leading to product recalls, damage to brand reputation and financial losses. Aseptic inline sampling allows for early contamination detection by monitoring microbial activity and other spoilage indicators. By promptly identifying and addressing contamination issues, breweries can prevent the release of compromised products into the market.
- Reduced Downtime and Waste: Aseptic inline sampling can be used to isolate microbial activity, reducing the teardown of equipment and interruption to the brewing process. This streamlines beer quality control procedures and reduces downtime associated with troubleshooting.
- Accurate Samples for Improved Results: Aseptic inline sampling helps ensure accurate test results for making informed business decisions. Breweries can gain valuable insights into process trends, identify areas for improvement, and optimize brewing parameters through aseptic inline sampling, empowering breweries to enhance their processes and maintain consistent product quality continuously.
- Proactive Line of Defense is to Reduce Exposure to the Environment and Bacteriophage: Quality management for bacteriophage requires a multifaceted approach and due diligence. Prevention, detection, and control are critical for avoiding the detrimental impacts of potential bacteriophage contamination on the quality of fermented products. Aseptic inline sampling methods in a fermentation vessel allow the technicians to take samples and inoculate the product while minimizing the chance of introducing bacteriophage naturally present in the environment.
Have questions about aseptic and representative sampling?
Need a quote? We’ll respond to
your request quickly.
Looking for one of our global distribution partners near you?
Implementing Aseptic Inline Sampling in Breweries
QualiTru’s simple-to-use aseptic inline sampling system means that the sampling equipment has been sterilized, effectively reducing the risk of contamination in both the process and the sample. This minimizes the chances of inaccurate and unreliable test results and allows breweries to take a proactive approach in identifying potential risks, optimizing their processes, and enhancing product quality and brand recognition.
The following key application sites should be considered to implement inline sampling in breweries effectively. Click here to download a printable version of key application sites with QualiTru product recommendations.
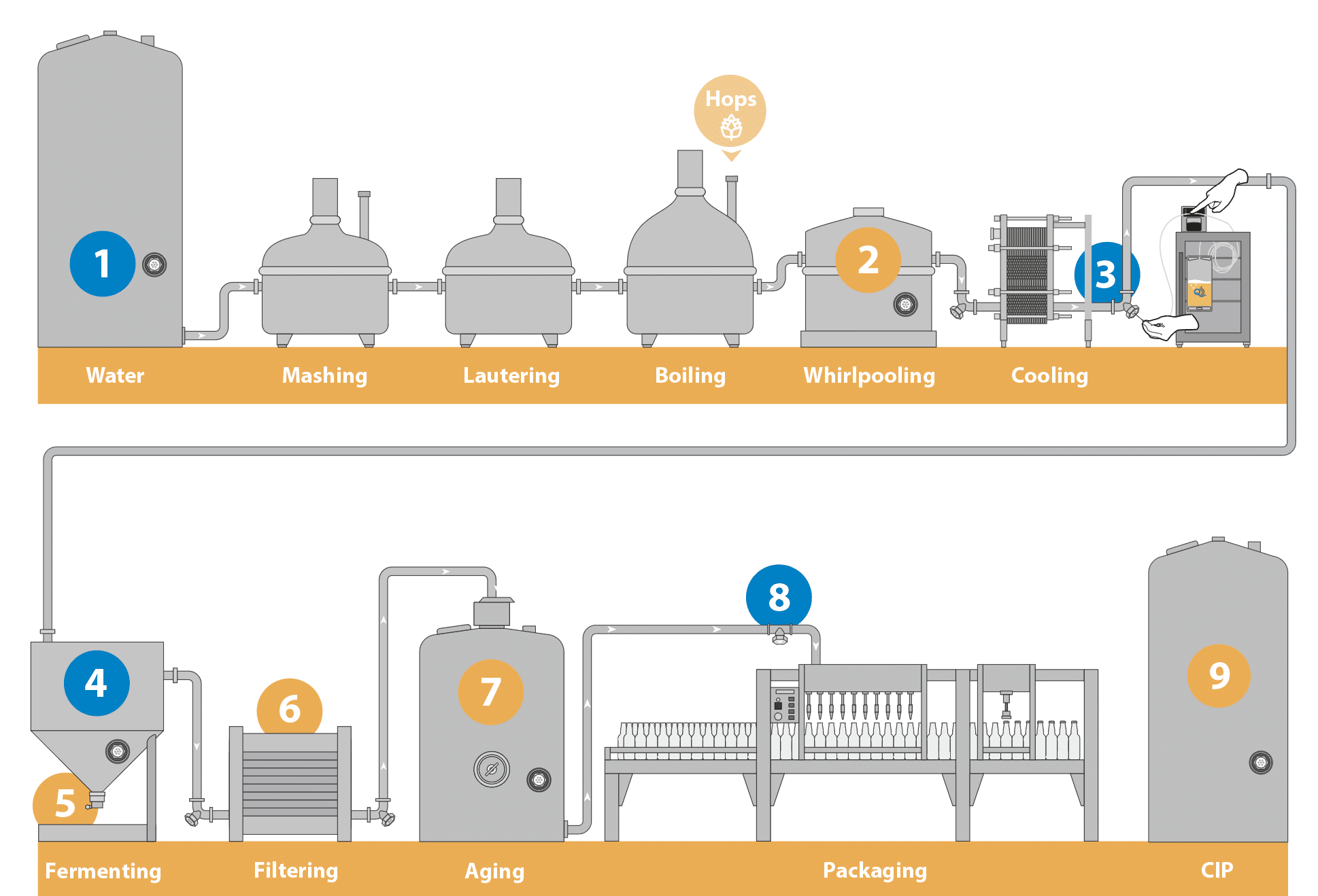
- Water Quality Verification
Test for water ions (calcium, magnesium, sulfate, sodium, chloride and bicarbonates) and microorganism counts. - Whirlpooling
Test wort for specific gravity, turbidity and microbiological contamination. - Wort Cooling and Peristaltic Pump
Test for microbiological contamination during knockout to avoid off-flavors. - Fermentation Tank
Sample at various times to check for fermentation progress to avoid ending fermentation early, resulting in off flavors. - Yeast Sampling
Test for accurate yeast cell count to avoid over or under-pitching into the wort. - Filtering
Sample before and after each piece of equipment as part of a system diagnostic function to check for alcohol, turbidity, yeast counts, and microbiological contamination. - Finished Product Sampling
Pull sensory and quality check samples throughout the beer maturation process. - Filler
Test the effectiveness of the system Clean in Place (CIP) process and obtain a product quality sample. - CIP Final Rinse
Sample the CIP rinse water to ensure the effectiveness of the process.
QualiTru Recommended Products to Get Started Using Inline Sampling to Monitor for Beer Contamination
Test for water ions (calcium, magnesium, sulfate, sodium, chloride and bicarbonates) and microorganism counts using the following QualiTru sampling system products:
- Inline Sampling
- TruStream7 Tri-Clamp Tee 2” (Part # 215147)
- TruStream7 Septa (Part # 110011)
- TruStream 250ml/18g (Part # 111450)
- Bulk Tank Sampling
- TruStream7 Recessed 4”x 2” (Part # 212123)
- TruStream7 Septa (Part # 110011)
- TruDraw® Sterile Single Sampler (Part # 112021)
Test for microbiological contamination during knockout to avoid off-flavors using the following QualiTru sampling system products:
- Inline Sampling
- TruStream7 Tri-Clamp Elbow 2” (Part # 213029)
- TruStream7 Septa (Part # 110011)
- TruStream 250ml/18g (Part # 111450) for a representative sample or the TruDraw® Sterile Single Sampler (Part # 112021) for a small, aseptic sample
Pull sensory and quality check samples throughout the beer maturation process using the following QualiTru sampling system products:
- Inline Sampling
- TruStream7 Tri-Clamp Tee 2” (Part # 215147)
- TruStream7 Septa (Part # 110011)
- TruStream 2L/18g (Part # 111730)
- Bulk Tank Sampling
- TruStream7 Recessed 4”x 2” (Part # 212123)
- TruStream7 Septa (Part # 110011)
- TruDraw® Sterile Single Sampler (Part # 112021)
Tap into the Benefits of Aseptic Inline Sampling in Breweries!
Aseptic inline sampling is a powerful tool for process monitoring and beer contamination control in breweries. By implementing aseptic inline sampling techniques, breweries can proactively monitor critical parameters, detect process deviations, and identify potential sources of contamination. This proactive approach detects deviations and contaminants, improving consistency and reducing downtime. Ensure high-quality beer production with the right equipment and data processes.
Complete this form to have one of our experts contact you to discuss implementing aseptic inline sampling in your brewery! Or call us at (651) 501-2337 or email [email protected] to learn more and/or to discuss your needs.



