Extended Shelf Life in Food Processing Starts Inside Your Equipment
Extended shelf life in food processing is often achieved through raw material quality, formulation, packaging, and temperature controlled distribution. However, recurring shelf-life loss often stems from the production system.
While many studies on production system related shelf-life loss focus on dairy plants, the causes of shelf-life loss and the resulting food waste apply broadly. One area where this can be significant is in closed system liquid food or beverage operations with less than optimal cleaning-in-place (CIP) systems. Persistent contamination can reduce quality and shelf life, cause product holds, and lead to avoidable downtime.
This blog is Part One of a two-part series on shelf-life protection and waste reduction in liquid food processing. Here, we examine how equipment design, microbial persistence, and cleaning effectiveness affect product performance. Part Two will address how these contamination issues lead to product loss, rework, and operational waste in food and beverage manufacturing.
Understanding how contamination develops inside equipment is the first step toward ensuring extended shelf life and preventing avoidable waste.
Top Five Causes of Reduced Shelf Life
Extending shelf life in food processing depends on controlling contamination within the production system. Research and industry investigations consistently point to a small set of recurring factors that drive reduced shelf life across liquid processing environments. Top five causes include:
|
Cause |
Where It Occurs |
How It Reduces Shelf Life |
What to Watch For |
|
Post-pasteurization contamination |
Downstream of pasteurization, including transfer lines, pumps, valves, and fillers |
Microorganisms enter the product stream after heat treatment and repeatedly contaminate batches |
Shelf-life variation between production runs |
|
Biofilm persistence |
Product-contact surfaces such as pipes, tanks, and gaskets |
Biofilms release microorganisms into product streams during processing |
Recurring contamination despite completed CIP cycles |
|
Hygienic design limitations |
Dead legs, low-flow zones, rough surfaces, gasket cracks |
Protected niches allow microorganisms to survive sanitation |
Persistent contamination in the same equipment locations |
|
Incomplete CIP performance |
Areas with insufficient temperature, chemical concentration, flow, or coverage |
Residues and microorganisms remain after cleaning cycles |
Shelf life declines despite routine sanitation |
|
Microbial enzyme activity |
Spoilage organisms such as Pseudomonas or spore-formers |
Proteases and lipases degrade product quality during storage |
Off-flavors, bitterness, texture changes |
These are only a few of the many factors that can quietly shorten product shelf life. Download our quick reference guide, Common Drivers of Reduced Shelf Life in Closed-System Liquid Processing, for a deeper look at where these risks occur and what to watch for across your process.
Why Extended Shelf Life Can Become Reduced Shelf Life
Shelf life rarely declines from a single failure. It usually erodes gradually due to low-level contamination that survives cleaning and sanitation.
In liquid processing, ineffective procedures can permit microorganisms to persist on equipment surfaces after CIP cycles and contaminate subsequent production runs. Over time, these microbes can cause:
- Off-flavors and bitterness
- Texture instability
- Gas formation
- Enzymatic degradation of proteins and fats
- Shortened shelf life across batches
These problems often appear inconsistently because the contamination source remains hidden inside the system.
Post-Pasteurization Contamination Limits Shelf Life
Heat treatment reduces microbial risks in product streams but contamination after pasteurization can still compromise finished product stability. Post-pasteurization contamination commonly originates from:
- Transfer lines
- Valves and pumps
- Holding tanks
- Fillers and packaging equipment
Persistent microorganisms in these areas can repeatedly contaminate product streams. In dairy and beverage systems, species like Pseudomonas produce enzymes that degrade quality during refrigerated storage, reducing shelf life.
How Biofilms Survive Cleaning-in-Place
Cleaning-in-place systems remove product residues and reduce microbial populations in equipment. However, microorganisms embedded within biofilms behave differently than free-floating cells.
Biofilms form when microorganisms attach to surfaces and produce protective extracellular layers. Once established, these structures can significantly increase resistance to cleaning and sanitizing agents (Sharma & Anand, 2002).
Even well-designed CIP cycles can have trouble removing contamination when biofilms grow in hard-to-reach areas of equipment. That’s why sanitation depends not only on chemicals but also on:
- Optimal cleaning temperature
- Correct chemical concentration
- Sufficient contact time
- Turbulent flow conditions
- Full physical coverage of product-contact surfaces
- Validated master sanitation schedules
- Competent sanitation teams
If these factors are not monitored accurately, contamination can persist inside the system and reduce shelf life.
Hygienic Equipment Design and Effective Master Sanitation Schedules Directly Affect Shelf Life
Good sanitation chemicals cannot make up for poor hygienic design or ineffective master sanitation schedules.
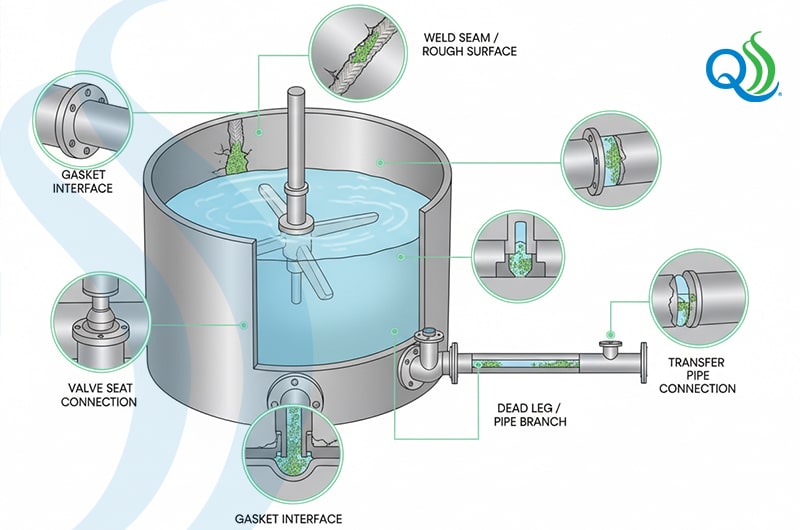
Across liquid processing operations, persistent contamination frequently develops in areas that are difficult to clean or only periodically get full breakdowns for cleaning and replacement. These include:
- Dead legs and low-flow zones
- Bad welds
- Scratched and rough stainless-steel finishes
- Gasket and seal interfaces
- Valve assemblies
- Pump connections
These features create protected niches where microorganisms can attach and survive standard sanitation cycles. Once established, biofilms can release microbes into the product stream repeatedly, causing persistent contamination.
Addressing hygienic design and master sanitation issues is often a key step to improving shelf-life stability.
Research Insight: Biofilms Can Survive Standard Cleaning-in-Place
Investigations of dairy processing equipment have recovered viable bacteria from product-contact surfaces even after CIP procedures.
In some cases, bacterial populations remained on surfaces at levels between 10³ and 10⁷ cfu/cm² following CIP cycles (Malek et al., 2012). These findings highlight how persistent biofilms can remain inside processing equipment even when sanitation protocols appear to function normally.
Once established, these microbial communities can repeatedly contaminate subsequent production runs and shorten product shelf life.
How In-Process Sampling Protects Shelf Life
Many processors rely primarily on finished product testing to confirm product quality. While this testing is important, it often fails to help explain or isolate the source of the contamination.
In-process aseptic sampling allows processors to collect representative samples directly from production lines at key control points or suspected trouble areas. Sampling during processing gives better insight into microbial conditions inside specific system components.
Strategically placed sampling points can reveal:
- Contamination emerging downstream of pasteurization
- Microbial growth developing inside the equipment
- Sanitation gaps between CIP cycles
- Gaps in master sanitation performance
- Microbial trends that may reduce shelf life
Since samples are collected directly from the process stream, in-process monitoring can identify contamination sources before the quality of multiple batches of finished product is affected.
This method offers earlier detection, clearer root-cause understanding, and better protection of extended shelf life than relying on end-product testing alone.
Four Critical Controls that Protect Extended Shelf Life In Food Processing.
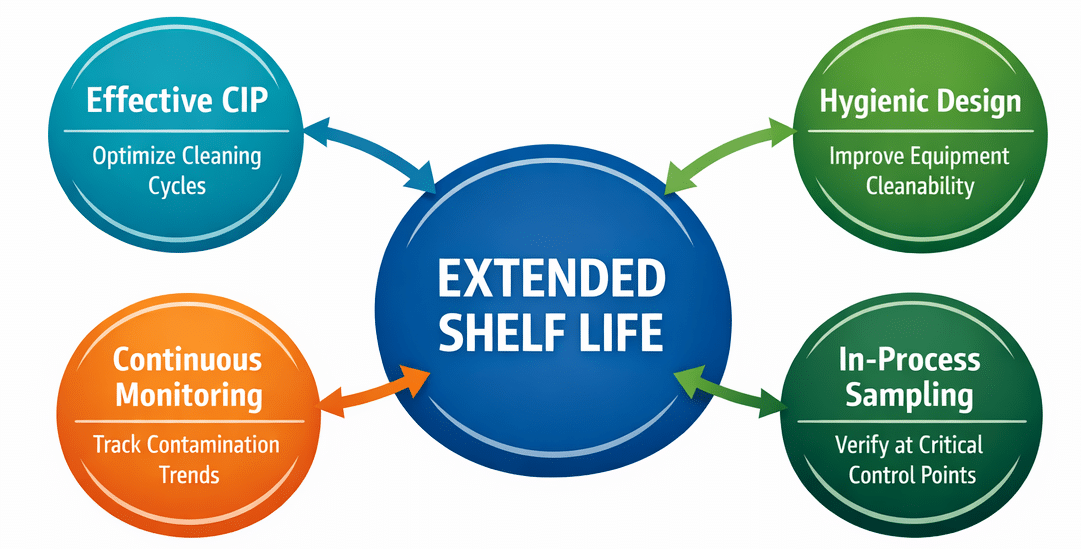
Extended Shelf Life Requires Eliminating Persistent Contamination
Achieving extended shelf life in food processing requires more than finishing sanitation cycles. It means knowing where contamination hides and making sure cleaning and sanitizing remove it effectively.
Processors can strengthen shelf-life protection by focusing on:
- Hygienic equipment design
- Data supported and effectively administered master sanitation
- Effective CIP performance
- Targeted monitoring of contamination hotspots
- In-process aseptic sampling at critical and targeted points in the production system
- Competence-instilling training for employees
When persistent contamination is eliminated, product stability improves. When contamination persists, the results are reduced shelf life, product inconsistencies, operational waste, and the resulting financial burden.
Part Two of this series, coming in April, will examine how shortened shelf life and contamination events translate into measurable operational waste in liquid food and beverage manufacturing. It will explore how microbial persistence drives product holds, rework, sanitation downtime, and ingredient loss, and how processors can identify hidden costs linked to shelf-life failures. Understanding how contamination control connects to waste reduction is key to improving both product quality and operational efficiency.
Improve Shelf Life by Identifying Hidden Contamination
Recurring shelf-life loss often indicates persistent contamination inside processing equipment.
QualiTru works with processors to investigate contamination sources, verify sanitation effectiveness, and implement monitoring strategies that detect microbial risks earlier in the production process. If shelf-life variability, microbial nonconformances, or sanitation challenges are affecting your operation, our team can help evaluate your system and recommend practical solutions.
Contact us at (651) 501-2337 or email [email protected] to learn more.
References:
Sharma, M., & Anand, S. (2002). Bacterial biofilms on food contact surfaces: A review.
F. Malek, Moussa-Boudjemacirc;a B., Khaouani-Yousfi F., et al (2012) Microflora of biofilm on Algerian dairy processing lines: An approach to improve microbial quality of pasteurized milk. https://doi.org/10.5897/AJMR11.1120
S. Cleto, Sónia Matos, L. Kluskens, M. Vieira (2012) Characterization of Contaminants from a Sanitized Milk Processing Plant. PLoS ONE. https://doi.org/10.1371/journal.pone.0040189
M. Kukhtyn, O. Berhilevych, K. Kravcheniuk, et al (2017) Formation of biofilms on dairy equipment and the influence of disinfectants on them. https://doi.org/10.15587/1729-4061.2017.110488