Proactive Biofilm Surveillance™
A New Approach to Contamination Prevention in Food and Dairy Processing
What is Proactive Biofilm Surveillance™?
Biofilms present a major challenge for food safety and processing. Proactive Biofilm Surveillance is a data-driven approach for anticipating and preventing biofilm formation in processing environments. By harnessing scientific insights to help identify biofilm hotspots. Quality assurance (QA) teams can focus on sampling, verifying sanitation, and preventing post-pasteurization contamination (PPC)—without disrupting production.
Once biofilms become established on surfaces and form their protective extracellular matrix, they are highly resistant to cleaning, sanitizers, and physical disruption. This results in persistent contamination that is difficult to trace, reduced shelf life, and serious food safety risks.
However, biofilm formation isn’t random. Research over the past several decades reveals that bacterial adhesion to surfaces follows predictable patterns. Gram-positive and Gram-negative bacteria attach to surfaces in different ways. By understanding how bacteria interact with various materials, QA and plant managers can anticipate biofilm formation and focus their surveillance efforts on high-risk areas.
The Science Behind Biofilm Attachment
Biofilm formation begins with bacterial adhesion to a surface. Initially, this process is reversible, with various forces competing to attract or repel bacteria. In watery environments such as milk, both bacterial and processing surfaces are typically negatively charged, which causes repulsion between them.
Key factors that influence early attachment include:
- Surface charge interactions: Negative charges on both cells and surfaces create electrostatic repulsion.
- Ionic conditions in the environment: Dissolved ions can change how strongly cells and surfaces repel or attract each other.
- Time and contact opportunity: The longer bacteria remain in contact with a surface, the greater the chance of attachment.
However, the presence of divalent cations, such as calcium and magnesium (Ca²⁺/Mg²⁺), can neutralize these charges, facilitating closer bacterial contact with the surface. These hardness ions reduce electrostatic repulsion between cells and surfaces, promoting initial attachment and ultimately biofilm formation.
In milk and similar matrices, calcium and magnesium can:
- Screen negative charges on both bacterial cells and processing surfaces
- Reduce electrostatic repulsion, allowing closer approach
- Promote stable initial adhesion that can progress to biofilm formation
Once bacteria make contact with the surface, they begin to deploy surface structures that help secure their attachment. For Gram-negative bacteria, this includes pili, fimbriae, and flagella, which physically aid in attachment. Conversely, Gram-positive bacteria often use adhesins, which are specialized proteins that help them bind to surfaces. In either case, attachment becomes irreversible when bacteria begin producing extracellular polymeric substances (EPS) that anchor them and protect the developing biofilm.
The real breakthrough is recognizing that not all surfaces are equal, and that different bacteria behave differently upon contact. This understanding is key to predicting where biofilms are likely to form.1,2
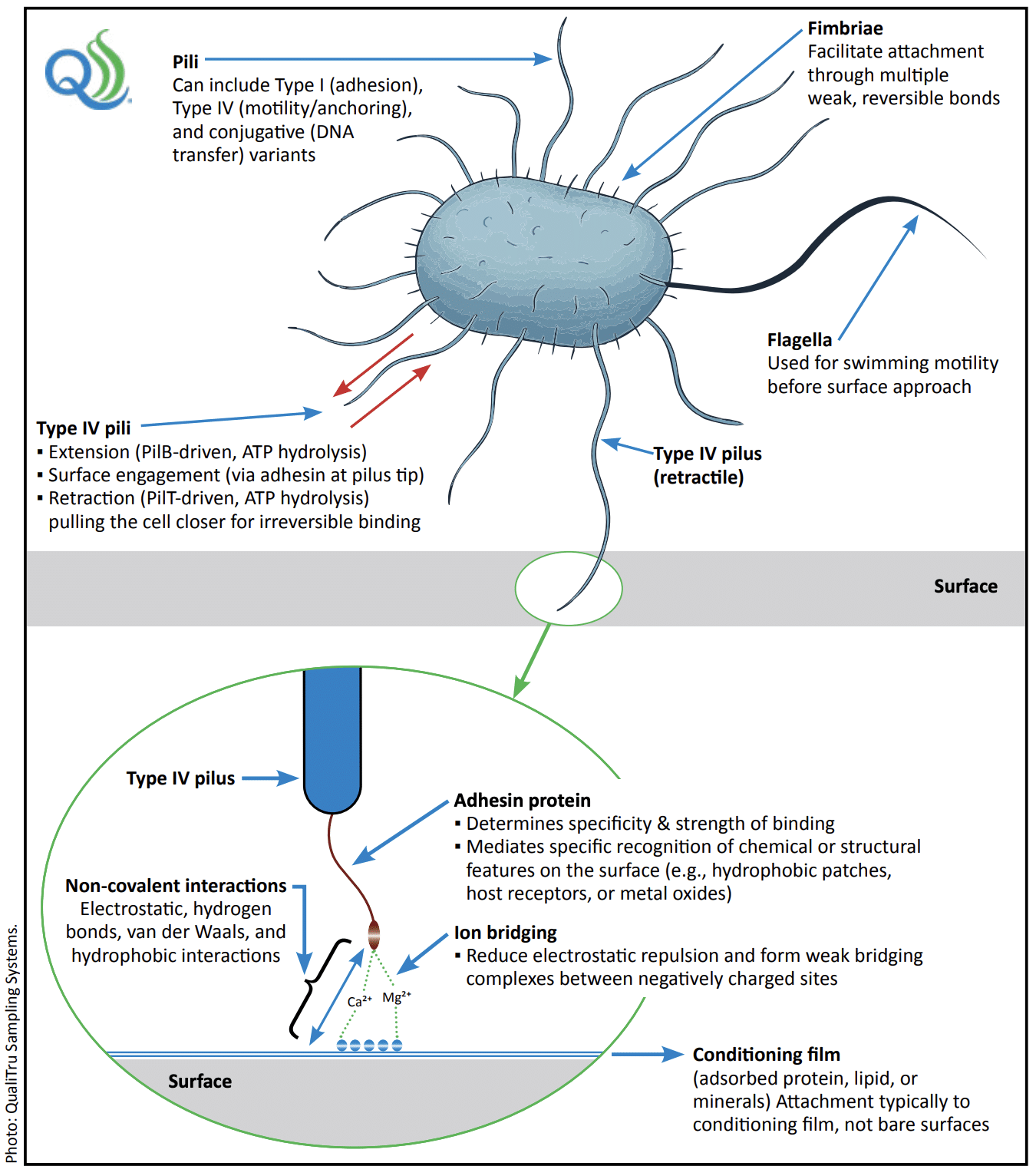
genera such as Escherichia coli, Salmonella enterica, Klebsiella pneumoniae, and Enterobacter spp. Type IV pili are retractile filaments that
extend from the cell surface to engage nearby substrates through a tip-associated adhesin. Both pilus extension and retraction are ATP-dependent processes driven by motor proteins: PilB, an ATPase that powers pilus extension, and PilT, an ATPase that drives retraction to pull
the cell into close contact for irreversible binding.
Attachment occurs primarily to a conditioning film of adsorbed proteins, lipids, or minerals
rather than to bare surfaces. Non-covalent forces, such as electrostatic, hydrogen-bonding, hydrophobic interactions, van der Waals forces,
and Ca²⁺/Mg²⁺ ion bridging, reduce charge repulsion and stabilize contact between the pilus tip and the surface. Fimbriae contribute to early,
weak, reversible attachment, while Type I pili promote stable adhesion that reinforces irreversible attachment, and conjugative pili mediate
DNA transfer between cells. Together, these surface structures coordinate the transition from reversible to irreversible adhesion that precedes
biofilm formation and subsequent maturation.
By contrast, Gram-positive bacteria such as Listeria monocytogenes lack pili entirely but employ surface-anchored adhesins, including the
Internalin family proteins (InlA, InlB, and related members), which are covalently attached to the cell wall by sortase enzymes. These adhesins
mediate direct contact with food-contact surfaces, contributing to their ability to colonize equipment, persist in processing environments, and
initiate biofilm formation through non-pilus mechanisms.
Surface Material is Critical to Biofilm Attachment
Food-contact surfaces vary in their ability to support biofilm formation based on how easily they are penetrated by water, surface energy, and texture. Stainless steel, while often considered hygienic due to its hydrophilic (water-attracting) properties, can still support biofilm formation. This is especially true in areas such as micro-scratches, welds, or sites where protein or fat residues accumulate.3
By contrast, hydrophobic (water-repelling) materials such as polytetrafluoroethylene (PTFE) and ethylene-propylene-diene monomer (EPDM) rubber attract bacteria that prefer non-polar surfaces. These surfaces exhibit low surface energy, which promotes stronger hydrophobic interactions and increases the likelihood of attachment by bacteria that thrive in hydrophobic environments. 3,4
How Gram-Positive vs. Gram-Negative Bacteria Attach
The structure of bacterial cells plays a key role in their ability to colonize surfaces. Gram-positive and Gram-negative bacteria differ in their cell structure and thus in their abilities to attach to different surfaces. This distinction is fundamental to understanding biofilm formation and the risks associated with different types of bacteria in food processing environments.
Guide to Gram-positive and Gram-negative bacteria differences and the practical impacts
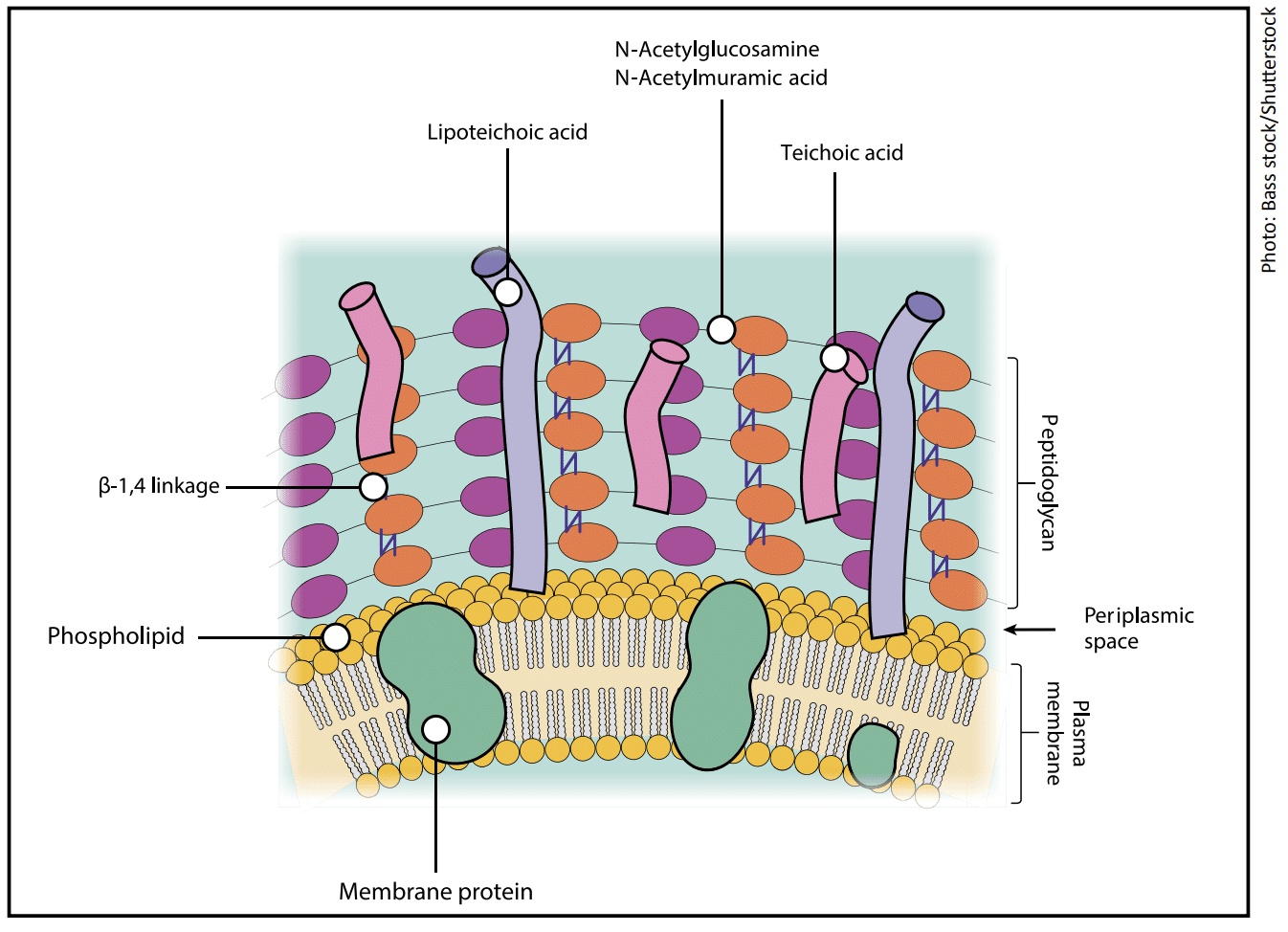
Gram-positive bacteria:
- Have a thick peptidoglycan wall, which is enriched with teichoic and lipoteichoic acids.
- These acids give the bacterial surface a strong negative charge, which makes it highly polar.
- The interaction between Gram-positive bacteria and hydrophilic surfaces is facilitated by divalent cations, such as calcium or magnesium, that are found in dairy and many liquid food products.
- These divalent cations act as bridges between the negatively charged bacteria and the hydrophilic surface, neutralizing the repulsive forces and allowing the bacteria to adhere.
- This bridging mechanism promotes electrostatic attraction and helps Gram-positive bacteria form stable attachments to hydrophilic surfaces like stainless steel or glass.4,5
Summary: Gram-positive bacteria are preferentially attracted to hydrophilic surfaces, such as stainless steel or glass, in the presence of divalent cations, such as calcium or magnesium, found in dairy and other liquid food products.
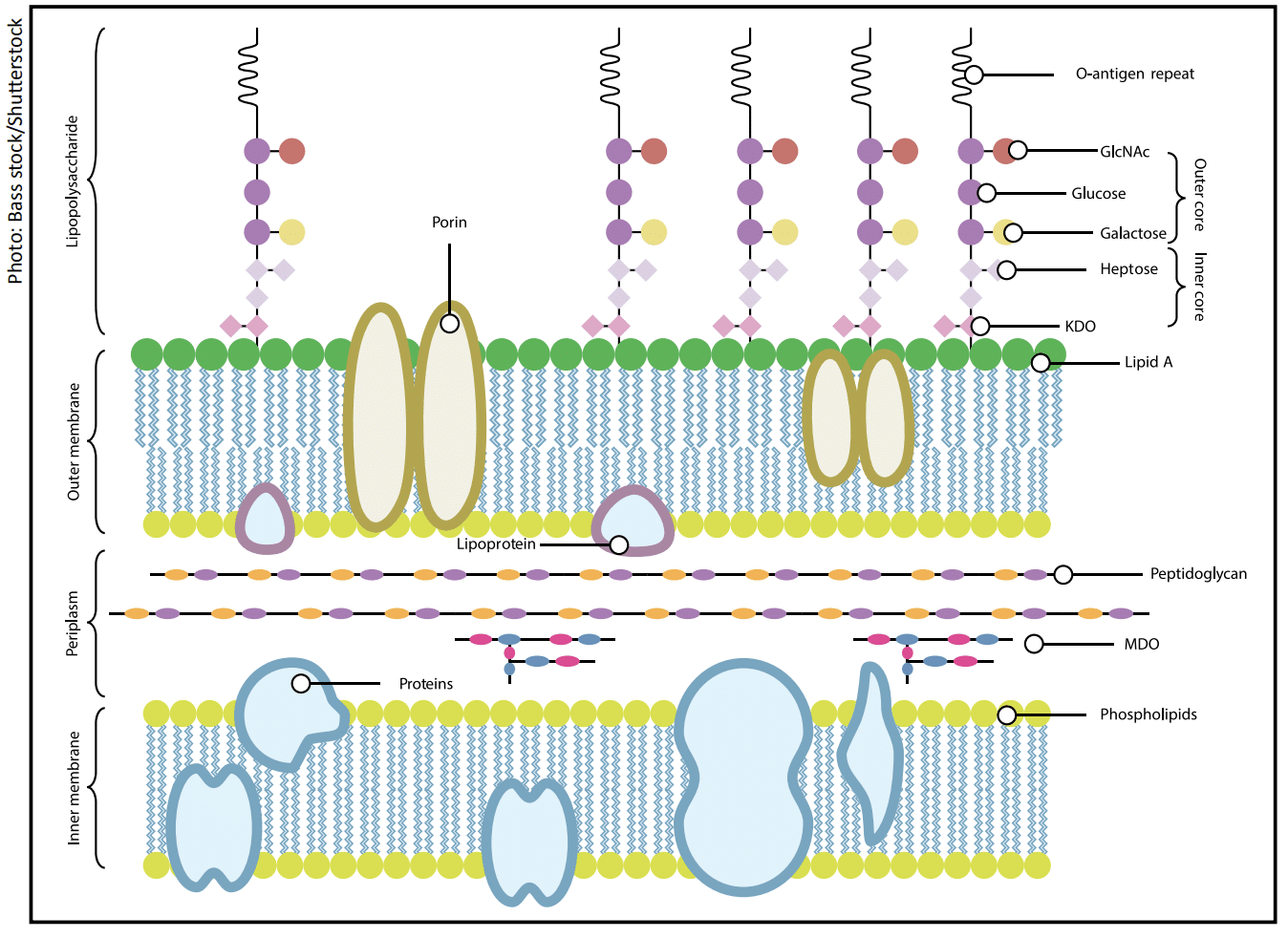
Gram-negative bacteria:
- Have a thinner peptidoglycan layer and an asymmetric outer membrane that contains lipopolysaccharides (LPS).
- LPS, molecules consist of lipid A, which is hydrophobic, and O-antigen chains, which are repeating units of monosaccharides.
- The O-antigen is hydrophilic and negatively charged, presenting an additional barrier to attachment.
- Divalent cations, in food, cannot penetrate deeply enough to reach the lipid A region of the bacterial membrane.
- Therefore, the ability to adhere to hydrophilic surfaces, such as stainless steel or glass, is impeded.
- However, when Gram-negative O-antigen chains are shortened or absent, the so-called rough form, the hydration layer is reduced.
- The bacteria are more likely to adhere to hydrophobic materials such as PTFE or rubber, as the exposed lipid regions now match the surfaces’ hydrophobicity.6-8
Summary: Gram-negative bacteria tend to adhere more readily to hydrophobic surfaces, such as PTFE or rubber.
A Strategic Approach to Predicting Biofilm Hotspots
By understanding biofilm formation mechanisms and surface properties of materials, food safety professionals can predict where biofilms are most likely to form. Proactive Biofilm Surveillance uses these insights to identify and monitor high-risk areas in food and dairy processing environments, focusing on areas most susceptible to biofilm development.
Hydrophobic vs. Hydrophilic Surfaces: Where Biofilms Start
|
Material |
Surface character |
Primary adhesion drivers |
Relative adhesion |
Conditioning film effect |
Sanitizer response |
Hotspots / notes |
QA/monitoring actions |
|---|---|---|---|---|---|---|---|
|
Stainless steel (SS)(AISI 304/316) |
Hydrophilic oxide layer; charge varies w/ pH/cleaners; smooth but microgrooved |
Electrostatics (strength ↑ w/ double-layer compression); conditioning films; microdefects; EPS– metal ion complexes |
Baseline (↓ PTFE/rubber; ↑ when residues present) |
Protein/fat films quickly mask the surface and ↑ ligands for adhesion |
Some sanitizers ↑ surface free energy and ↓ attachment; effectiveness depends on residues |
Imperfections, welds, scratches; EPS at periphery; micro-induced corrosion risk via Ca2+/Fe2+ complexes |
Prioritize sampling at welds/defects/Δ flow; verify residue removal |
|
PTFE and similar polymers |
Highly hydrophobic; very low surface free energy; smooth |
Hydrophobic interactions; rapid protein /lipid film formation |
Highest among listed materials (often > SS, glass, rubber) |
Readily adsorbs proteins/lipids; films drive attachment despite native hydrophobicity |
Larger sanitizer effect vs. Rubber/ SS; films detach more readily |
Gaskets, valves, tubing, belts; initial attachment is high even when clean |
↑ surveillance; validate sanitizer contact time; inspect for film removal and reformation |
|
Rubber (e.g., EPDM) |
Moderately– strongly hydrophobic; rougher; microcrevices |
Hydrophobic interactions; rapid protein film formation; microdefects |
Intermediate (PTFE > rubber > SS ≈ glass when clean) |
Rapid, persistent protein films (casein/whey) facilitate attachment |
Less effect vs. PTFE; porosity and elasticity can limit efficacy |
Gaskets, seals; niches resist clean-in-place (CIP); reservoir for recolonization |
Short inspection & replacement intervals; aggressive CIP and validate; aggressive monitoring |
|
Glass |
Hydrophilic; high surface free energy; smooth, but scratches are possible |
Electrostatics at certain pH/ionic strength; conditioning films overcome resistance |
Lower when clean → rises with films of scratches |
Milk proteins readily adsorb → ↑ attachment to levels comparable to other materials |
Effective when films are minimal; scratches can harbor cells |
Scratched zones; legacy equipment; less common as food contact today |
Check for scratches; ensure film removal; decommission damaged pieces |
Key Strategies for Identifying and Managing Biofilm Hotspots
- Match the Contaminant to the Surface
- If Gram-positive bacteria are identified, focus on hydrophilic surfaces such as stainless steel, glass, ceramic, polished concrete, or aluminum (often coated with oxide layers). These surfaces attract electrostatic and hydrogen-bonding interactions that favor attachment of Gram-positive bacteria.
- For Gram-negative bacteria, attention should shift to hydrophobic surfaces, including PTFE-coated surfaces, polyurethane, EPDM rubber, polypropylene, polyethylene, and silicone seals and gaskets. These materials support the hydrophobic interactions characteristic of Gram-negative bacteria, which increase their propensity for biofilm formation.
- Factor in Process Chemistry
- Many food products, byproducts, and processing ingredients contain divalent cations, such as calcium and magnesium, which can bridge bacterial and surface charges to reduce repulsion and facilitate bacterial attachment. Low-flow regions where these ions can accumulate should be closely monitored, as they are ideal sites for biofilm formation.
- Watch for Residues and Conditioning Films
- Even when surfaces appear clean, microscopic layers of proteins, fats, or minerals may remain. These conditioning films can mask the natural resistance of clean surfaces and create new binding sites for bacteria, allowing them to colonize areas that would otherwise resist attachment. Sanitation and surveillance programs must account for these films to ensure surfaces are thoroughly cleaned. Protein, fat, and mineral soils can seed an EPS matrix and accelerate quorum-sensing-driven biofilm development—even on “clean” surfaces.
- Use Data-Driven Monitoring
- Routine environmental sampling and testing should be aligned with predicted hotspot locations. Use data-driven monitoring to prioritize inspections and trend aerobic plate counts (APC), coliforms, and spores to confirm control and catch contamination trends early. Adjust cleaning schedules based on product type, surface material, and historical contamination trends.
Practical Applications for Proactive Biofilm Surveillance
Applying these strategies helps plant managers implement proactive strategies for biofilm prevention and detection. Here are a few practical steps QA managers can take to predict and prevent biofilm hotspots:
- Material Selection Awareness
- Recognize the surface characteristics of food-contact materials. Hydrophobic polymers promote Gram-negative colonization, while hydrophilic surfaces tend to attract Gram-positive bacteria. Understanding the role that material chemistry plays in biofilm formation can help incorporate this knowledge into equipment design and maintenance.
- Process Chemistry Control
- Regularly monitor water hardness, pH, and sanitizer formulations. Small shifts in these factors can alter bacterial attachment potential by influencing surface charge and ionic strength, underscoring the need to maintain control over these variables.
- Enhanced Cleaning Verification
- Use various surface testing techniques to ensure that surfaces are free of organic residues that may form conditioning films. Use aseptic inline (closed-system) sampling to verify the absence of lingering contaminants or biofilms and to track results over time. These verification procedures are critical to confirming that cleaning protocols are effective.
- Targeted Environmental Mapping
- Use Gram classification data from routine microbiological isolates to prioritize inspections of high-risk areas. Align sampling frequency with risk levels based on surface characteristics, process conditions, and historical data. Focus on receiving lines, raw storage, pre-/post-pasteurization, balance tanks, valves/gaskets/dead legs, and fillers—then refine placement based on isolates data and trend results.
- Adaptive Sanitation Protocols
- Tailor cleaning protocols to different surface types. PTFE and rubber may require more prolonged exposure or different sanitizer chemistries than stainless steel. Regularly rotating sanitizers can help prevent resistance and enhance cleaning effectiveness.
This approach supports Hazard Analysis and Critical Control Points (HACCP) and Food Safety Modernization Act (FSMA) preventive controls by strengthening your environmental monitoring program (EMP) and clean-in-place (CIP) verification.
Know the Surface, Own the Outcome
Biofilms are persistent and costly, but they are not unpredictable. By understanding the scientific principles behind bacterial attachment, QA managers and plant executives can more effectively predict biofilm hotspots and isolate troublesome biofilms when they arise.
By aligning sanitation strategies with the biology of biofilm formation, water quality, dairy, and food plants can shift from reactive cleaning cycles to a more proactive and efficient approach. Proactive Biofilm Surveillance focuses on preventing biofilms from taking hold and quickly locating them when they appear. The results are fewer holds and reworks, faster root-cause analysis, and more consistent product quality.
Ultimately, it is about safeguarding product safety and ensuring brand integrity.
Visit our White Papers and Articles page to learn more about biofilms and QualiTru.
Proactive Biofilms Surveillance Q&A
Q: What is Proactive Biofilm Surveillance?
A proactive approach that uses known attachment behaviors of Gram-positive and Gram-negative bacteria and surface properties (hydrophobic/hydrophilic, surface energy, texture) to predict where biofilms will form and focuses monitoring on high-risk areas.
Q: Which materials are highest risk for different contaminating organisms?
Hydrophilic surfaces (e.g., stainless steel, glass) tend to attract Gram-positive bacteria (cation “bridging” can facilitate attachment). Hydrophobic polymers (e.g., PTFE, EPDM, silicone) tend to favor Gram-negative attachment.
Q: How do conditioning films and divalent cations increase attachment risk?
Hardness ions (Ca²⁺/Mg²⁺) can reduce charge repulsion and help cells approach surfaces. Residual proteins/fats/minerals can form conditioning films that mask inherent resistance and create new binding sites. Both elevate biofilm risk.
Q: Where should we focus sampling and verification?
Align environmental/inline sampling with predicted hotspots: materials that match the organism’s risk profile, low-flow or ion-accumulation zones, and areas with residues/conditioning films; then adjust based on isolate data and historical trends.
Q: How does Proactive Biofilm Surveillance support continuous control?
It transforms surface and microbial insights into actionable monitoring strategies—using ongoing data collection, inline sampling, and sanitation verification to prevent biofilm establishment before it becomes a contamination issue.
Map your surfaces. Align organisms. Verify with aseptic sampling. Contact us at (651) 501-2337 or email [email protected] and start safeguarding your product and brand with Proactive Biofilm Surveillance!
We offer two convenient ways to request a quote:
Request for Quote (RFQ)
Visit our product pages to select items and submit an RFQ. A QualiTru representative will follow up promptly with pricing and ordering information.
Custom Quote Form
Use our Custom Quote Form to provide details about your specific operation and the challenges you’re facing. A QualiTru representative will follow up with more information.
References:
1. Bos R, Van der Mei HC, Busscher HJ. Physico-chemistry of initial microbial adhesive interactions—its mechanisms and methods for study. FEMS Microbiol Rev. 1999;23(2):179–230.
2. Van Loosdrecht MCM, Lyklema J, Norde W, Zehnder AJB . Influence of interfaces on microbial activity. Microbiol Rev. 1987;54(1):75–87.
3. Gammudi BA, El Musrati IM, Bourgheya AM, El Tawil AA, Telegdi J. (2008). Microscopic study of bacterial attachment on AISI 304 and AISI 316 stainless steel surfaces. Int J Integr Biol. 2008;4(2):72-79.
4. Barnes LM, Lo MF, Adams MR, Chamberlain AH. Effect of milk proteins on adhesion of bacteria to stainless steel surfaces. Appl Environ Microbiol. 1999;65(10):4543-4548.
5. Brown S, Santa Maria JP Jr, Walker S. Wall teichoic acids of Gram-positive bacteria. Annu Rev Microbiol. 2013;67:313-336.
6. Jennings LK, Storek KM, Ledvina HE, Coulon C, Marmont LS, Sadovskaya I, Secor PR, Tseng BS, Scian M, Filloux A, Wozniak DJ, Parsek MR, Howell PL. Pel is a cationic exopolysaccharide that cross-links extracellular DNA in the Pseudomonas aeruginosa biofilm matrix. Proc Natl Acad Sci U S A. 2015;112(36):11353-11358.
7. Pratt LA, Kolter R. Genetic analysis of Escherichia coli biofilm formation: Roles of flagella, motility, chemotaxis and type I pili. Mole Microbiol. 1998;30(2):285–293.
8. Houry A, Briandet R, Aymerich S, Gohar M. Involvement of motility and flagella in Bacillus cereus biofilm formation. Microbiology. 2010;156(4):1009–1018.