Food Manufacturing Waste Reduction: How Hidden Contamination Drives Product Loss
In food and beverage manufacturing, product waste usually starts long before it becomes visible. Often, by the time a batch is placed on hold, a shipment is rejected, or product keeping quality/shelf life drops, the problem has been developing inside the processing system for days or weeks.
In Part One of this series, we looked at how persistent contamination inside equipment can reduce product shelf life. Biofilms, design flaws, and incomplete cleaning all increase microbial survival, allowing microorganisms to contaminate new batches repeatedly (Sharma & Anand, 2002; Kukhtyn et al., 2017).
Part Two focuses on how shelf-life reduction is not the only result of hidden contamination. Ingredient loss, product holds, rework, lost time on equipment, sanitation downtime, and product withdrawals and recalls are other factors that can be linked to persistent contamination. Understanding how microbial contamination leads to operational waste is key to improving efficiencies and managing food manufacturing waste reduction.
When Reduced Shelf Life Becomes Operational Waste
Reduced shelf life is a visible symptom of persistent contamination, as it affects the finished product and customer satisfaction. However, the microbial contamination that impacts product life has symptoms that, while less connectable or quantifiable, can disrupt the entire production process.
Common outcomes include:
- Product holds pending microbial testing
- Batch rejection due to spoilage indicators
- Increased sanitation cycles between runs
- Production downtime during contamination investigations
- Early product expiration during distribution
- Loss of customer confidence
- Unmet Key Performance Indicators (KPIs), including customer complaints and sustainability
- Increased chemical and water use
- Increased cost for sanitation inventory
- Increased wastewater load on wastewater systems and municipal agencies
- Increased fines from municipalities for nutrient load of influents
These disruptions, and their after-effects, increase product, time, and efficiency losses, while worsening food manufacturing waste issues.
Many facilities view waste events as separate issues instead of costs related to contamination and indicators of deeper,contamination-related problems within the system.
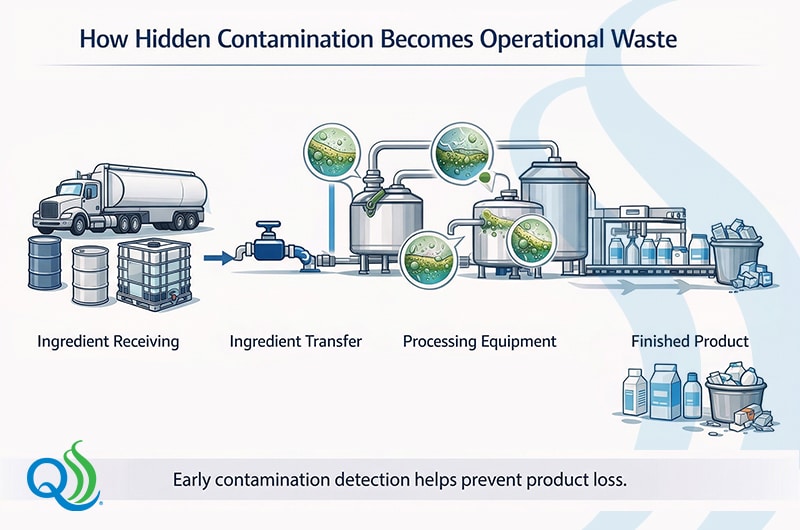
Waste Cascade Map: How Hidden Contamination Becomes Waste
Hidden contamination rarely causes immediate waste. Instead, it triggers a chain of operational problems that gradually lead to product loss, downtime, and reduced efficiency.
|
What Starts the Problem |
What It Triggers |
What It Looks Like in the Plant |
Waste Impact |
|---|---|---|---|
|
Persistent contamination inside equipment (biofilm) |
Low-level microbial release into product streams |
Inconsistent microbial test results and unexplained shelf-life variation |
Increased product rejection and rework |
|
Post-process contamination |
Product holds pending investigation |
Finished product lots are waiting for microbial results |
Delayed shipments and increased inventory risk |
|
Biofilm survival after cleaning-in-place (CIP) |
Repeat sanitation cycles |
Additional cleaning and sanitation verification before production can resume |
Production downtime and increased labor costs |
|
Contaminated incoming ingredients |
Contamination entering multiple batches |
Microbial growth detected across several production runs |
Raw material loss and batch disposal |
|
End-product testing as the primary detection method |
Late discovery of contamination |
Product already produced, packaged, or distributed before detection |
Highest waste (and waste cost) per contamination event |
These cascading effects show why contamination control is central to food manufacturing waste reduction. Detecting contamination only after finished product testing limits options to prevent product loss and reduce the incidental costs.
Monitoring contamination during ingredient verification, sanitation validation, and in-process sampling help interrupt this cascade before waste occurs.
Where Waste Often Begins: Hidden Contamination in Equipment
Microorganisms persisting inside equipment can periodically release cells into the product stream. If this occurs after heat treatment, contamination can quickly compromise product stability (Cleto et al., 2012).
Once contamination begins affecting batches, processors may experience:
- Shortened product shelf life
- Inconsistent microbial test results
- Increased product holds or withdrawal/recalls
- Recurring sanitation interventions
Because these events are intermittent, identifying the contamination source is difficult. It is important to know that persistent microbial reservoirs often exist in:
- Gasket interfaces
- Valve assemblies
- Dead legs and low-flow zones
- Scratched or rough stainless steel surfaces
- Improperly cleaned transfer lines
These niche areas can allow microorganisms to survive sanitation cycles and repeatedly contaminate product streams (Kukhtyn et al., 2017), as well as form biofilms in other areas. Systematic troubleshooting is required to identify contamination sources once biofilms have developed.
The Hidden Cost of Shelf-Life Failures
Shelf-life failures often manifest as customer complaints, product returns, or expired stock but their real cost extends beyond the product’s supply chain. Contamination that shortens shelf life leads to several operational problems, including:
- Product Loss: Batches may be discarded due to microbial counts, spoilage, or regulatory compliance issues.
- Associated costs: Raw materials (including shipping and handling), labor (including time overages), waste disposal.
- Rework and Ingredient Waste: Unreleased products may require reprocessing or disposal, wasting raw materials.
- Associated costs: Raw materials, equipment operation, utilities, operational efficiencies, labor, product shipment delays, customer confidence, and waste disposal.
- Production Downtime: Contamination investigations and additional cleaning can delay production schedules.
- Associated costs: operational efficiencies, cleaning and sanitation inventories, additional product loss, product shipment delays, and customer confidence.
- Increased Sanitation and Testing: Facilities may increase sampling and testing frequency or intensify sanitation to better control contamination risks.
- Associated costs: operational efficiencies, labor, cleaning and sanitation inventories, water, utilities, and municipality fees.
Together, these events contribute to measurable challenges in food manufacturing waste reduction and sustainability initiatives and increased operating costs across production operations.
Why End-Product Testing Alone Cannot Prevent Waste
Many facilities rely heavily on finished product testing to verify quality and safety. While important, this often detects contamination only after production is affected, thus reducing a facility’s ability to control and reduce losses.
By the time contamination appears in finished products, the product is already made, packaged, and possibly shipped. Processors can then only hold, rework, or discard them.
Preventing and reducing waste requires spotting microbial risks earlier in the process and conducting effective investigations, root cause analyses (RCA), and corrective and preventive action (CAPA) to resolve the issue.
Consistent monitoring for contamination, at multiple points within the production system, offers better insight into microbial persistence and aids investigations and RCA in finding the source of the contamination.
Root Cause Investigation: When Shelf Life Declines
When product shelf life declines or microbial counts begin trending upward, identifying the root cause quickly is key to preventing product loss and operational waste. The following investigation pathway can help processors narrow down potential contamination sources.

Detecting Contamination Earlier in the Production System
Early detection strategies focus on monitoring microbial conditions within the production environment rather than waiting for finished product failures.
Several monitoring approaches can help processors identify contamination before it affects product stability:
- Consistent monitoring of product lines through multipoint inline/in-process sampling
- In-process aseptic sampling at critical control points
- Environmental monitoring of processing areas
- Verification of sanitation effectiveness
- Targeted microbial testing of equipment surfaces
- Trending and cross-trending microbial results and related activities
Inline or in-process sampling lets processors collect representative samples directly from production lines. Monitoring microbial conditions within the process stream helps detect contamination trends earlier and aids in identifying root causes.
This proactive approach helps processors address contamination before it reduces shelf life or causes product loss.
Waste Prevention Starts with Ingredient Integrity
Hygienic ingredient sampling before production helps detect contamination early and prevents microbial risks from spreading across batches. Closed-system sampling technologies enable facilities to verify ingredient quality while protecting product integrity.
Ingredient Sampling Can Prevent Waste Before Production Begins
Contamination risks do not always originate inside the processing line. Out-of-specification ingredients are another source of product waste. Opening ingredient packaging for sampling prior to production can also lead to ingredient degradation and waste.
Many facilities receive ingredients packaged in drums or totes, such as syrups, oils, concentrates, and liquid additives.
This packaging can make it difficult to sample and still maintain ingredient integrity. Unsealing and removing drum caps or bung for sampling exposes the product to the environment. This sampling process can introduce air, causing oxidation and microbiological contamination into the container. Contaminated or degraded ingredients entering the process can cause reduced product quality, microbial growth, shorter shelf life, and product loss across multiple batches.
Closed-system sampling methods, on the other hand, allow processors to verify ingredient quality without exposing product to airborne contamination. For example, our TruStream™ 3 NPS Drum Sampling Cap with an integrated septum and 4” side hole needle, allows hygienic sampling without opening the drum.
These systems help processors verify supplier Certificate of Analysis (COA), assess ingredient quality, monitor for food fraud, maintain product integrity, and reduce contamination risk before production.
You can learn more about our TruStream™ 3 NPS Drum Sampling Cap, which allows hygienic ingredient sampling directly through a sealed drum port.
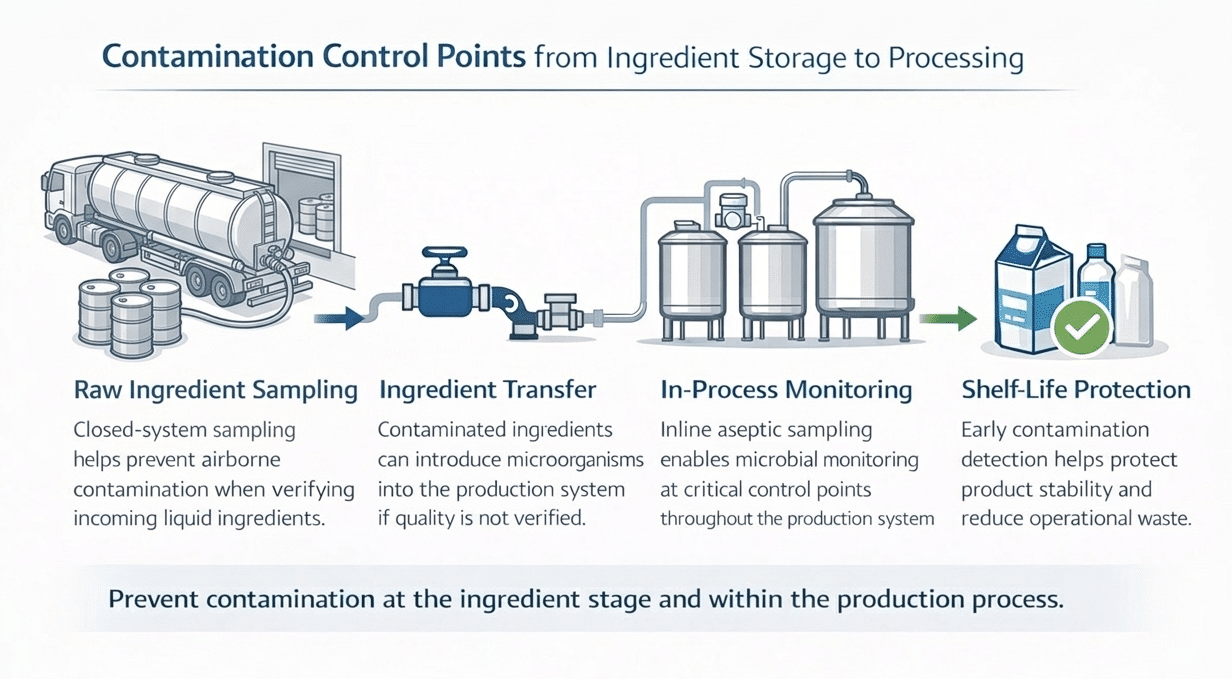
Connecting Shelf-Life Protection and Waste Reduction
Reducing food manufacturing waste and sustainability challenges often begins with improved contamination control.
Facilities that successfully reduce waste typically focus on several key areas:
- Hygienic equipment design that minimizes contamination niches
- Effective CIP programs that remove residues and microbial growth
- Monitoring strategies that track contamination trends
- In-process sampling to detect microbial risks earlier
- COA verification and monitoring for food fraud
These practices help processors identify contamination pathways before they cause product instability, production downtime, or waste.
Strengthening Contamination Control with Technical Expertise
Identifying root causes of repeated contamination can be challenging. Many facilities face complex systems where cleaning, equipment design, and microbial behavior interact. Technical support that delivers systematic and focused investigation can help.
QualiTru Technical Services offers expert support to processors for assessing contamination risks, improving monitoring, and enhancing shelf-life protection. Our technical specialists help facilities:
- Identify contamination hotspots in processing systems
- Evaluate sanitation and CIP effectiveness
- Develop targeted monitoring and sampling strategies
- Improve microbial visibility across production operations
- Educate personnel to help reduce contamination and waste
Combining equipment expertise, process knowledge, and educational training with microbial monitoring help processors improve contamination control and reduce operational waste.
Contact us at (651) 501-2337 or email [email protected] to learn more about how our team supports contamination control and process monitoring.
References:
Cleto, S., Matos, S., Kluskens, L., & Vieira, M. (2012). Characterization of contaminants from a sanitized milk processing plant. PLoS ONE.
Kukhtyn, M., Berhilevych, O., Kravcheniuk, K., Horyuk, Y., Semaniuk, N., & Horiuk, V. (2017). Formation of biofilms on dairy equipment and the influence of disinfectants on them. Eastern European Journal of Enterprise Technologies.
Sharma, M., & Anand, S. K. (2002). Bacterial biofilms on food contact surfaces: A review. Journal of Food Science.