Micro Testing for Post-pasteurization Contamination (Part 3)
This is a guest post written by Neil Bogart, Executive Area Technical Support Coordinator of NA Food & Beverage for Ecolab Inc. He is a highly respected adviser in the food and beverage industry, providing troubleshooting, technical support, cleaning, and sanitizing solutions to the dairy, food, beverage, and bottled water markets. See Author Bio at the end of the post.
Part 3 Topic: Micro Testing for Post-pasteurization Contamination
The first article in this series, Raw Milk Dairy Micro Testing, focused on Gram-negative bacteria in raw milk with the Preliminary Incubation (PI) Count and Fresh Standard Plate Count (SPC) tests. The second article, The Effects of Gram-positive HRSF Bacteria on Fluid Milk Shelf Life, discussed another classification of bacteria that was not known as a major issue in fluid milk processing prior to 2001. In this third installment, I would like to discuss micro testing for post-pasteurization contamination. When discussing post-pasteurization, once the milk has passed the divert valve, all the equipment and product are now on the pasteurized side of the process. You can receive quality raw milk, have low Gram-negative bacteria counts, and low heat-resistant spore formers (HRSF), and then one little issue on the pasteurized side of the plant can negate all that work done on the raw side.
What are potential sources of post-pasteurization contamination and how do we find them?
Seemingly little issues like a bad gasket, valve or stem O-ring, air blow, and/or improperly cleaned and sanitized equipment are all potential harboring sites, and/or potential causes for contamination. Of these, gaskets and air-blows tend to be the most common ones when diagnosing quality issues. That’s why it is critically important that gaskets are on the Preventive Maintenance (PM) program and that the maintenance schedule is strictly followed, and that air-blows receive the same strict adherence on the Master Sanitation Schedule (MSS). If all plants had effective gasket and air-blow controls, post-pasteurization contamination issues would be significantly reduced, and I would likely be out of a job.
Using micro testing to determine issues with processing equipment.
Micro testing to identify and isolate contamination issues within the processing equipment is also essential and effective in combination with the PM and MSS programs, provided that the correct micro tests are selected, and that sampling and testing are done at the right locations.
The need for aseptic inline sampling cannot be overstated, as accurate micro testing for post-pasteurization contamination results are dependent upon having accurate and reliable sampling. Aseptic inline sampling is critical throughout the post-pasteurized side of the process, including at the discharge of the high temperature short time (HTST), in and out of silos, and just before the fillers. The maintenance of these sampling ports is also critically important (see QualiTru’s Standard Operating Procedure (SOP)).
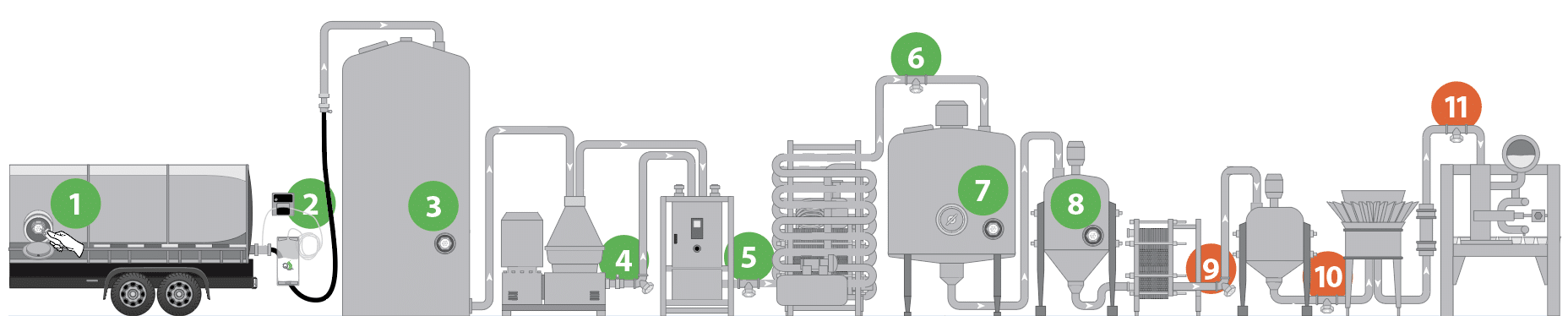
We need to remember that the lab’s customers are the sanitation, production, and maintenance teams. The lab needs to get the most useful data to these teams in the fastest manner so they can be efficient and timely at developing and implementing corrective actions for micro counts that are out of specification. We will discuss this further in the fourth and final article of this series.
Micro testing for post-pasteurization contamination requires answering three key questions.
- First, which classifications of bacteria are of concern?
- Second, what is the desired shelf life of the product?
- And finally, which tests can most quickly enumerate the organisms we are concerned with based on the desired shelf life?

Below is a breakdown of micro testing for post-pasteurization contamination options with some pros and cons of available tests for identifying them, based on my experience.
Gram-negative pathogens
Coliforms are a non-pathogenic subset of the family Enterobacteriaceae. Without specifically testing for a pathogen, we can look at a non-pathogenic indicator and know that there is a potential for a pathogenic presence in the product. Coliforms do not survive pasteurization; therefore, coliform detection is an indicator of unsanitary conditions in food processing or unsanitary water quality. If there are coliform counts in the finished product, we need to focus sampling and testing on the discharge of the HTST forward to the filler.
There is one significant drawback to the Fresh Coliform Test. If damaged coliforms are present, they may not show up on the Fresh Coli Test. For this reason, I recommend that the Stress Coli Test be used in conjunction with the Fresh Coli Test.
It is important to note the following if you are micro testing an acidified or acidic product.
The acid can inhibit the growth of coliforms. You may have coliforms in the product, but they will not grow out in the acidified environment. You must first neutralize the acid in the product before testing for coliforms or you can utilize the Charm® Peel Plate® C-CD Bacterial Test. It has a neutralizer built into the plate, so you do not have to do a neutralization of the product before plating.
The Stress Coli Test utilizes Dr. C.K. Johns’ understanding that bacteria stressed by processing need to repair themselves before plating in order to be successfully enumerated. Again, the stressing of a product is another way of saying preliminary incubation (PI).
The pasteurized milk sample is held at 70°F (21°C) for 18-24 hours, based on sample size, and then plated on VRB agar with a cap.
The Fresh Coli Test is your legal test, and the Stress Coli Test is your insurance test. These two tests used together can effectively identify issues before they become uncontrollable and measure above the legal requirements. The legal requirement in the U.S.A is <10cfu/mL on Fresh Coli Test.
The HR-1 (stress test for post-pasteurization contamination) Test is a quick and simple qualitative test that helps pinpoint coliform and other Gram-negative post-pasteurization contaminating bacteria that cause most post-pasteurization safety and quality problems. The HR-1 Test will flag products that have potential contamination issues within 22 hours of production. The test accomplishes this by stressing the product and allowing the growth of coliform and other Gram-negative bacteria while strongly inhibiting the growth of any competing Gram-positive bacteria. If these contaminating bacteria are present, they will grow during the test. The color of the product-reagent solution will turn from purple to pink and then finally to white. If a color change is noticed after stressing the sample for 22 hours at 89.6°F (32°C) the sample is considered “positive”.
An important distinction to remember is that the HR-1 can help determine Gram-negative post-pasteurization contamination within 22 hours of bottling or sampling, whereas the Stress Coli Test takes up to 48 hours, requiring 24 hours for PI and 24 hours of incubation after plating. The HR-1 is a qualitative test, whereas the Stress Coli Test is quantitative.
Gram-negative spoilage organisms
Decades ago, when plants were trying to achieve a ten-day shelf life, Moseley’s Laboratory introduced a testing method based on the belief that the majority of microorganisms causing shortened shelf life were Gram-negatives post-pasteurization contamination, usually located at the filler. The lab preincubated a sample of pasteurized milk off a filler at 45°F (7.2°C) for five days. Then it was plated for Total Plate Count (TPC). As shelf life extension demands increased due to improved cleaning and process changes that were driven by Moseley test results, the laboratory determined the need to increase the five-day PI to seven days. When they did this in the late 1970s to early 1980s, it significantly increased the identification of psychrotrophic Gram-negatives. As a result, in the 1980s, Standard Methods transitioned to a 7-Day Moseley.
If you follow Moseley’s method intent, the PI at 45°F (7.2°C) was developed to understand the psychrotrophic bacteria counts at roughly 50% of the desired shelf life. Therefore, I question the continued use of the 7-Day Moseley by the fluid industry if the majority of the industry is pushing for 18, 21, and out to 30 days code on conventionally pasteurized milk. You would have to increase your PI to 50% of the desired shelf life, respectively.
If we make this transition to the intent of the method, I would challenge how the data that you had an issue with 11, 13, or 17 days ago helps you. What is the chance you remember what happened in the plant five or seven days ago? However, I will state that some customers have had success at correlating their Moseley counts to their desired shelf life once they transitioned to 50% of their desired shelf life.
This test is also referred to as Aerobic Plate Count (APC) or TPC. It is the measure of the total population of viable aerobic organisms in a given sample.
One negative for a fresh SPC is that just as in the Fresh Coli, you can have stressed and damaged bacteria present that may not show up on the fresh SPC Test. For this reason, I recommend the Stress SPC Test over the Fresh SPC Test.
Based on the scientific work of Dr. C. K. Johns and Dr. Moseley, the Virginia Test was developed so we could see viable bacterial counts just after pasteurization. Again, the milk is heated up, cooled down, and moves through numerous pumps and valves. Bacteria are injured or stressed. So, the milk is held at 70°F (21°C) for 18 – 24 hours and then plated on TPC agar. The plate is then incubated at 89.6°F (32°C) for 48 hours, or for better association with Psychrotrophs, it can be incubated at 69.8°F (21°C) for 72 hours. The choice is dependent on the process and desired shelf life.
The test is more selective towards Gram-negatives, but Gram-positives can also grow out. Typically, if you are having high counts on this test, they are Gram-negative, and if low counts they are Gram-positive, based on the lag phase for both bacterium types. However, I have had a few cases with too numerous to count (TNTC) on these tests and they were Gram-positive. This is where Gram-staining can help with differentiation because we would treat a Gram-negative issue differently than a Gram-positive one.
This test allows you to begin building a correlation at the plant to predict potential shelf life. But it is not the only test to run for shelf life prediction.
The HR-2 (presumptive shelf life) Test is a simple qualitative test used to help predict the shelf life of fluid dairy products within 24 to 72 hours of production. It has been shown that much of the post-pasteurization shelf life problems are caused by Gram-negative bacteria such as Pseudomonas; however, towards the end of code, the poor shelf life may also be caused by Gram-positive bacteria. As many of these bacteria will survive pasteurization, Gram-positive bacteria may or may not be a post-pasteurization contamination issue. If the HR-2 Test is pointing at Gram-positive bacteria as the major issue after consistent 48 to 72-hour failures, then the HR-3 can be used to isolate the Gram-positives.
The Stress PPC Test (Alles, A. A., 2016) is a newer test developed by the Milk Quality Improvement Program, Department of Food Science, at Cornell University. The test utilizes the already run Stress Coli Test, so there is no need to plate another microplate. Follow the procedure for the Stress Coli Test and read the plate after 24 hours of incubation. Then place the plates back into the incubator for another 24 hours and again count the plates. The count is representative of Gram-negative post-pasteurization contamination. In the SPC Test, Stress SPC, and coli tests, Pseudomonas do not grow out well, though we know Pseudomonas are a major source of PPC. The new Stress PPC Test has shown a good enumeration of Pseudomonas in the samples.
Gram-positive spoilage organisms
Gram-positive organisms can grow on the 7-Day Moseley, SPC, Stress SPC, and HR-2 tests. However, it is hard to differentiate the Gram-positives from Gram-negatives without Gram-staining the growth on the plates. The HR-2 has the advantage of starting to differentiate the contamination bacterium based on the time of failure.
Gram-positive HRSF spoilage organisms
This is a quantitative HRSF micro test developed for the review of psychrotolerant, mesophilic, and thermophilic spore counts by Cornell (Kent, D. J., 2016). Obtaining the psychrotolerant counts takes up to ten days. To speed up the psychrotolerant test turn time, Consolidated Lab Services (CLS) modified the test. The lab pasteurized sample is held at 55ᵒF (13ᵒC) for 48 hours and then plated for the determination of the psychrotolerant load. Utilizing the expedited psychrotolerant HRSF test, we found that the initial action level on individual farm loads, raw milk loads, high temperature short time (HTST) balance tank, and HTST discharge samples is ≥100 cfu/mL.
Read more about Gram-positive HRSF testing in my second blog post, The Effects of Gram-positive HRSF Bacteria on Fluid Milk Shelf Life.
The HR-3 Test is a screening test for thermoduric/psychrotrophic Gram-positive spore-forming microbial contamination. The test is a quick and simple qualitative stress test that helps pinpoint in raw or pasteurized milk the Gram-positive bacteria that will survive pasteurization and possibly adversely affect the product’s finished quality and shelf life. Within 14 hours, the HR-3 Test will flag products that have potential contamination issues. The test accomplishes this by stressing the product and allowing the growth of specific thermoduric/psychrotrophic Gram-positive microbes. The color of the product-reagent solution will turn from purple to pink and then finally white if Gram-positive contaminating bacteria are present. The color change is recorded after stressing the sample for 14 hours at 89.6°F (32°C). Following the incubation period, performing a microscopic examination is necessary on positive samples to further quantify the Gram-positive bacteria. So far, articles one, two, and three have discussed raw and pasteurized milk Gram-negative bacteria contamination, Gram-positive, and Gram-positive HRSF bacteria, and the common tests that can be used to enumerate them. In the final article, we will bring all these tests together to develop an effective micro-program to support your desired shelf life and methods of processing.
There’s still more to understand about micro testing for post-pasteurization contamination and effective micro-programs.
So far, articles one, two, and three have discussed raw and pasteurized milk Gram-negative bacteria contamination, Gram-positive, and Gram-positive HRSF bacteria, and the common tests that can be used to enumerate them. In the final article, we will bring all these tests together to develop an effective micro-program to support your desired shelf life and methods of processing.
Additional resources related to dairy micro testing for post-pasteurization contamination:
Free on-demand webinars:
Monitoring Microbial Contaminations
Previous blog posts:
Raw Milk Dairy Micro Testing (Part 1)
The Effects of Gram-positive HRSF Bacteria on Fluid Milk Shelf Life (Part 2)
Psychrotrophic Bacteria in Pasteurized Milk—Spoilage, Testing, and Line Sampling
Maintaining the Quality of Pasteurized Fluid Milk
Why is Aseptic Inline Sampling Critical to Process Monitoring?
Training
Why Process Monitoring & Aseptic Sampling Information Sheet
Additional Videos
Finding solutions for contamination issues can be challenging, frustrating, time-consuming, and sometimes costly! If you have a contamination issue you are having trouble solving or would like to discuss a proactive process monitoring plan, contact us at (651) 501-2337 or email [email protected] to have one of our experts contact you.
Reference: (1) Alles, A. A., Wiedmann, M., & Martin, N. H. (2018, June 18). Rapid detection and characterization of postpasteurization contaminants in pasteurized fluid milk. Journal of Dairy Science. https://www.journalofdairyscience.org/article/S0022-0302(18)30571-X/fulltext (2) Kent, D. J., K. Chauhan, K. J. Boor, M. Wiedmann, and N. H. Martin. 2016. Spore test parameters matters: Mesophilic and thermophilic spore counts detected in raw milk and dairy powders differ significantly by test method. J. Dairy Sci. 99:5180-5191. (https://doi.org/10.3168/jds.2015-10283)